 /periodic-table-of-elements-680789917-58ea3e903df78c5162f92b6f.jpg)
Going from left to right in the periodic table the electropositivity decreases.Going top to bottom down the group, the electronegativity decreases because the atomic number of an element increases down the group and thus the distance between the outermost electrons and the nucleus increases and the atomic radius.Since, electronegativity is opposite of electropositivity, we can say that the periodic trends of an electropositive element is always opposite to the periodic trends of an electronegative element.īelow we have given the periodicity of the element 
The periodic trends of the electropositive elements can vary depending upon various factors such as the ionisation energy of an element, the metallic character of the element, distance between the outermost electron and the nucleus of an element and the effective nuclear charge on the valence electrons. Periodic Trends in the Electropositive Element Most electronegative element are Fluorine or Chlorine Most electropositive elements are Francium or Cesium 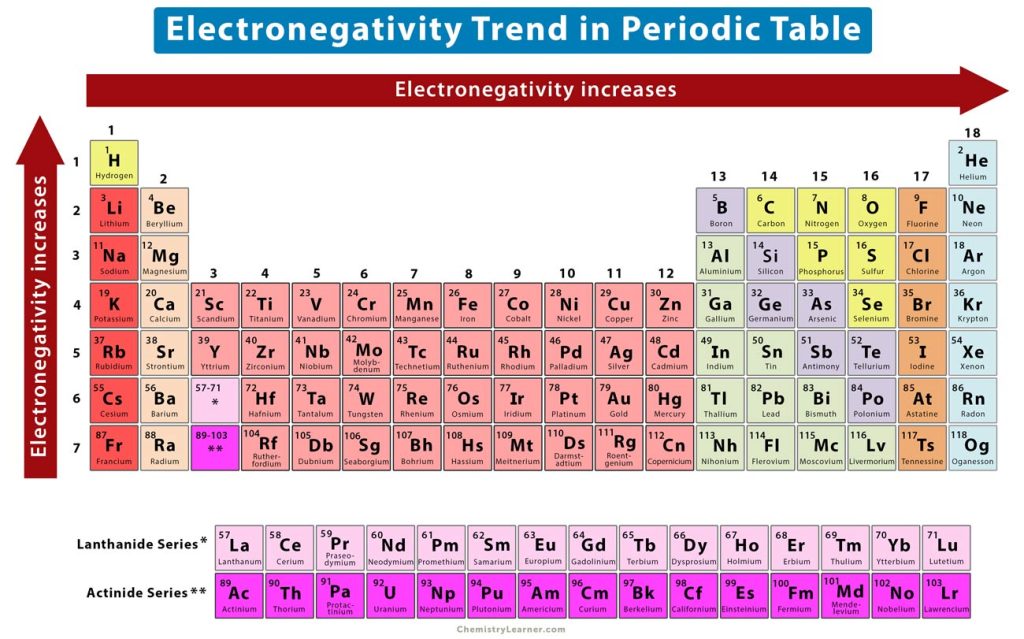
Shown by mostly metals such as alkali and alkaline earth metals It forms negatively charged anions by accepting one or more electrons It forms positively charged cations by removing one or more electrons It has the ability to attract electrons towards itself It is ability of an element to gain or accepts electrons It refers to the ability of an element to lose electrons in a chemical reaction Magnesium Chloride is useful to treat and prevent the low amount of magnesium in the blood. Therefore from the above mentioned example we can say, the electropositive element Magnesium and the electronegative element Chlorine combined to form an ionic salt named Magnesium Chloride or MgCl2. For example, Magnesium (Atomic Number :12) which has two electron in its outermost orbit lose its electron to form a positively charged cations in order to achieve the nearest noble gas electronic configuration whereas Chlorine (Atomic Number :17) a highly negative elements accepts the two electron to form a positively charged anion in order to complete its stable octet. The electropositive elements lose electrons to electronegative elements to form an ionic compound. The higher the electronegativity character of an element, the lower the electropositive elements and vice versa. Alkali metals or alkaline metals.Įlectropositivity is opposite to the term of electronegativity in which an element accepts an electron from another element to form a negatively charged anion. The ability of an atom to form a cation is mostly shown by the metallic elements of the periodic table i.e. The term Electropositive is generally defined as the tendency of an atom to release its electron to another atom to form a positively charged cation. Periodic trends in the Electropositive element.Difference between the Electropositive and Electronegative. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
